Clinical Research
WHO THIS IS FOR
Clinical Operations and Procurement Teams Running Phase I-IV Trials
This solution is built for teams that:
-
Coordinate multiple specialized vendors across trial sites without a unified system
-
Need to standardize processes and documentation across different clinical sites
-
Struggle to track clinical trial spend and commitments in real time
-
Need to prevent supplier overpayment through verified milestone and invoice matching
-
Must maintain complete audit documentation for regulatory inspection readiness

THE PROBLEM THIS SOLVES
Fragmented vendor management delays trial timelines and patient enrollment
Clinical trials require coordinated execution across multiple sites and specialized service providers. Yet fragmented systems for managing clinical vendors, each with different coordination processes, documentation requirements, and reporting formats, create delays that push out enrollment timelines and increase costs.
Enter: end-to-end clinical trial support beyond simple buying assistance.
.webp?width=1529&height=2000&name=coordination-complexity@2x%20(1).webp)


.webp)
HOW THE SOLUTION WORKS
From fragmented to coordinated
-
Automate
Establish compliant workflows for all trial-related services leveraging pre-negotiated agreements. Automated intake ensures protocol requirements are captured upfront.
-
Coordinate
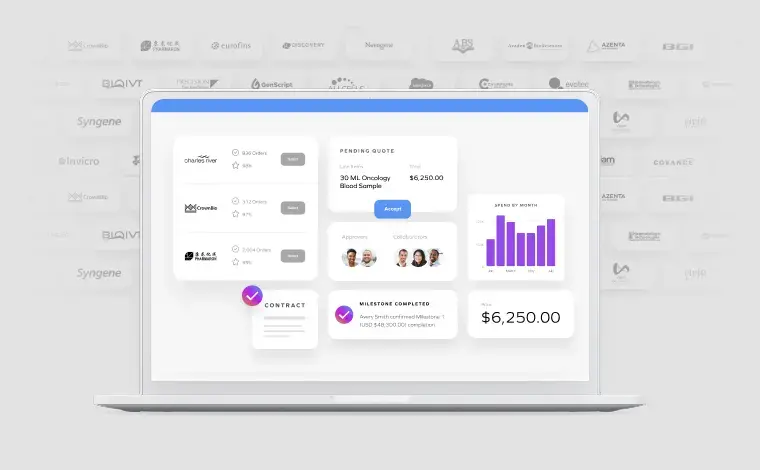
Centralized platform coordinates supplier activities across all trial sites. Real-time visibility into vendor performance, milestone completion, and budget utilization.
-
Document
Comprehensive audit trails track all supplier interactions, compliance verification, and payment processing—ready for regulatory inspection.
HOW THE SOLUTION WORKS
From fragmented to coordinated

Establish compliant workflows for all trial-related services leveraging pre-negotiated agreements. Automated intake ensures protocol requirements are captured upfront.

Centralized platform coordinates supplier activities across all trial sites. Real-time visibility into vendor performance, milestone completion, and budget utilization.
.webp)
Comprehensive audit trails track all supplier interactions, compliance verification, and payment processing—ready for regulatory inspection.


WHAT MAKES THIS DIFFERENT
We orchestrate science, not transactions
Domain Expertise and Data Model
We have decades of scopes, performance records, pricing benchmarks, and workflow patterns that exist nowhere else, allowing you to move beyond your own transactional data to power your decisions.
Purpose-Built for Science
Unlike generic tools, we understand protocol-level requirements, scientific expertise evaluation, and regulated R&D workflows.
Complete Orchestration
We don't just connect you to suppliers. We orchestrate sourcing, compliance, contracting, payment, and performance analytics in one unified platform.
Unified Legal Framework
One Master Services Agreement covers thousands of suppliers—eliminating the need to negotiate individual contracts, CDAs, and MTAs for each relationship or project.
Global Scientific Ecosystem
Curated supplier partners, top pharma companies in the world, along with emerging biotech and a team of scientists—all working together to orchestrate scientific breakthroughs.
PROOF
Measurable outcomes for clinical operations
6.5x
Faster project start time
5 Weeks
Reclaimed per scientist per year, returned to research instead of administration
21%
Cost saving on life science purchasing through optimized sourcing and compliance*