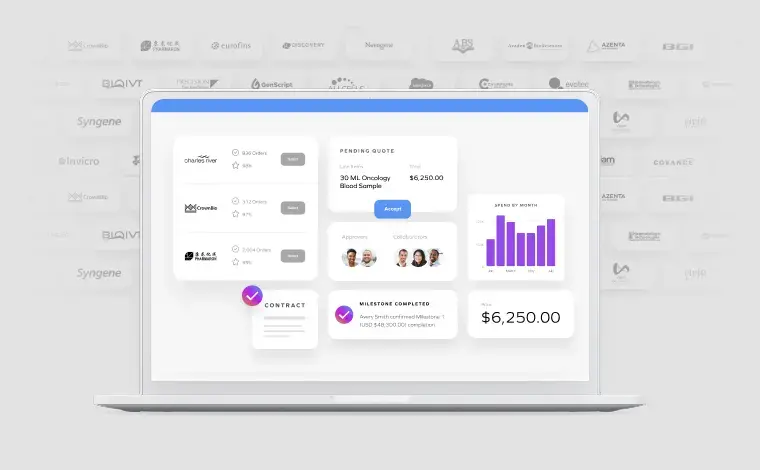
One Agreement.
Thousands of Supplier Relationships.
Legal Review Done Once.
Science Exchange gives legal and contracts teams the intelligent infrastructure to standardize supplier agreements at scale, apply consistent IP and data protection provisions across every engagement, and remove legal review from the critical path for every project start.
Predictive contract coverage that protects every supplier relationship, before risk appears.
Proactive compliance built into the framework, not retrofitted after the fact.
Precise audit documentation that stands up to regulatory inspection, without the scramble.
THE PROBLEM
A bespoke contract for every supplier is not a legal strategy. It is a liability.
At R&D scale, with dozens of active supplier engagements running simultaneously and new ones beginning every week, the contract-by-contract review model consumes legal capacity without improving risk posture. Each bespoke agreement is an opportunity for IP protection provisions to vary, data handling clauses to be inconsistent, and liability terms to reflect whoever reviewed the contract that particular quarter.
The result is a supplier contract record where standard terms are not actually standard, compliance provisions depend on recency and reviewer, and audit readiness requires weeks of manual compilation.
The culprit: a contract-by-contract approach to supplier management designed for occasional vendor relationships, applied to a continuous, high-volume R&D supply chain.

Every engagement is a new negotiation
Each new supplier relationship triggers a full contract review cycle. At R&D scale, that cycle runs continuously and occupies legal capacity that should be focused on higher-stakes work.
Consistent terms are not consistently applied
IP protection provisions, data handling clauses, and liability terms vary across the supplier contract record depending on when each agreement was reviewed and who reviewed it. The record is spread across hundreds of individual files.
Legal is on the critical path for every project start
Because each new supplier engagement requires a contract review, legal sits between the project request and the project start. That position creates schedule pressure on legal teams and downstream delays for science teams.
THE SOLUTION
Intelligent contract governance built for the way life sciences procurement actually works.
Science Exchange replaces the per-engagement contract review cycle with a unified legal framework that covers every supplier relationship under one pre-approved Master Services Agreement (MSA).
- Unified MSA Framework
- Built-In Regulatory Compliance
- Centralized Compliance Record
- Legal Framework
What you get: Your IP protection, data handling, liability, and compliance provisions apply to every supplier relationship automatically, without a separate review for each one.
A single pre-approved MSA covers all suppliers on the Science Exchange network. Legal terms are accepted at network entry. Standard provisions apply consistently across every engagement. Bespoke negotiation per supplier is not required.
What you get: Your GDPR, HIPAA, and FDA requirements are incorporated into the contract framework, not retrofitted into agreements after the fact.
Data protection provisions, regulatory compliance requirements, and audit trail obligations are built into the MSA. Legal does not need to review each agreement individually for regulatory compliance. When requirements change, updates happen at the framework level.
What you get: Your complete contract and compliance documentation is in one governed location, accessible for inspection without manual compilation.
Every supplier agreement, certification status, and compliance verification is maintained in the platform throughout the supplier relationship. Regulatory or internal audit preparation does not require manual assembly across email threads and file systems.
The Science Exchange MSA is a master agreement covering all supplier engagements on the platform. IP ownership, data handling, liability, and regulatory compliance provisions are standard in every supplier relationship. Legal review happens once at the framework level. This is an executed, enforceable agreement applied at network scale.
What you get: Your IP protection, data handling, liability, and compliance provisions apply to every supplier relationship automatically, without a separate review for each one.
A single pre-approved MSA covers all suppliers on the Science Exchange network. Legal terms are accepted at network entry. Standard provisions apply consistently across every engagement. Bespoke negotiation per supplier is not required.
What you get: Your GDPR, HIPAA, and FDA requirements are incorporated into the contract framework, not retrofitted into agreements after the fact.
Data protection provisions, regulatory compliance requirements, and audit trail obligations are built into the MSA. Legal does not need to review each agreement individually for regulatory compliance. When requirements change, updates happen at the framework level.
What you get: Your complete contract and compliance documentation is in one governed location, accessible for inspection without manual compilation.
Every supplier agreement, certification status, and compliance verification is maintained in the platform throughout the supplier relationship. Regulatory or internal audit preparation does not require manual assembly across email threads and file systems.
The Science Exchange MSA is a master agreement covering all supplier engagements on the platform. IP ownership, data handling, liability, and regulatory compliance provisions are standard in every supplier relationship. Legal review happens once at the framework level. This is an executed, enforceable agreement applied at network scale.


WHY SCIENCE EXCHANGE
Built for legal teams who support R&D operations at scale.
Every capability is designed for the specific risk profile of high-volume, regulated R&D supplier management, not adapted from a general commercial contract framework.



-
Regulatory Provisions That Update at the Framework Level
When GDPR requirements, HIPAA obligations, or FDA audit standards change, updates apply to the framework. Legal teams are not responsible for identifying which existing agreements need to be individually amended and tracking that amendment process.
-
IP and Data Protection That Are Not Negotiable Per Supplier
IP ownership and data handling provisions are not subject to per-supplier negotiation. They are standard terms that apply to every relationship on the network. There is no category of supplier for which these provisions are reduced or absent.
-
Compliance Monitoring That Does Not Require Legal Follow-Up
Supplier certifications and insurance are tracked automatically with expiration alerts built into the platform. Legal teams are not responsible for monitoring renewal cycles or following up with suppliers on documentation

When GDPR requirements, HIPAA obligations, or FDA audit standards change, updates apply to the framework. Legal teams are not responsible for identifying which existing agreements need to be individually amended and tracking that amendment process.

IP ownership and data handling provisions are not subject to per-supplier negotiation. They are standard terms that apply to every relationship on the network. There is no category of supplier for which these provisions are reduced or absent.

Supplier certifications and insurance are tracked automatically with expiration alerts built into the platform. Legal teams are not responsible for monitoring renewal cycles or following up with suppliers on documentation


FOR EMERGING BIOTECH
Solutions for every stage of your growth
Science Exchange gives early-stage biotech teams the R&D operations infrastructure they can't yet afford to build. So your scientists stay focused on what the company was created to do. Access the network, the negotiating leverage, and the operational backbone of an enterprise R&D operations team without the headcount, the contracts, or the overhead.

Resources
Procurement Strategy & Digital Transformation
Buyer’s Guide Capability Deep Dive: Reimagining Financial Workflows & Analytics in External R&D
Ready to remove legal review from the critical path for every supplier engagement?
The contract-by-contract review model has a compounding cost: in legal capacity, in project start delays, and in the inconsistent risk posture that accumulates across hundreds of individually negotiated agreements. Science Exchange provides the framework that makes one review sufficient for all of them.