In our previous article, we introduced the shift from orchestration software to what we define as Intelligent Infrastructure for Science, a more connected, system-level approach to managing external R&D.
At the center of that model are six core capabilities that, together, enable organizations to operate as a cohesive system rather than a collection of tools.
In this piece, we're taking a deeper look at the first two capabilities: Marketplace & Supplier Management and Project/Study Management. These are foundational. They define how work enters the system and how it is executed once it begins. And in most organizations today, they are also where fragmentation is most visible and most costly.
Most R&D organizations still restart supplier qualification from scratch with every engagement—here's why that's holding science back.The First Challenge: Supplier Access and the Cost of Starting from Scratch
For most R&D organizations, every new external engagement still begins the same way: a scientist identifies a need, procurement initiates supplier discovery, compliance begins qualification, and legal starts contract review.
Even in organizations that have invested in orchestration tools, much of this process still happens across disconnected systems and stakeholders. Supplier information lives in one place. Qualification documentation in another. Contracts are negotiated separately. Communication is scattered across inboxes.
The result is not just inefficiency, it is repetition. Every engagement effectively resets the process.
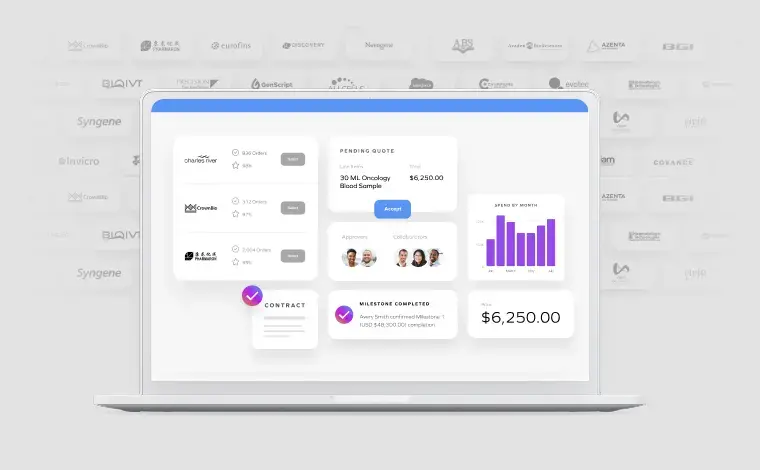
Capability 1: Marketplace & Supplier Management
A modern R&D operations system does not treat supplier engagement as a one-time transaction. It treats it as a persistent, reusable capability.
At its core, this means providing:
- Access to a pre-qualified supplier network
Suppliers are vetted in advance across technical, compliance, and operational criteria, eliminating the need to repeat qualification work for every engagement. - Standardized contractual frameworks
Master service agreements and pre-approved terms remove the need for full contract negotiation each time, compressing onboarding timelines from months to days. - Self-service access within governance guardrails
Scientists can identify and engage suppliers directly, while procurement and compliance maintain control through predefined policies and workflows. - Persistent supplier performance data
Institutional knowledge – delivery timelines, quality metrics, responsiveness – is captured and reused across projects, preventing both redundant evaluation and repeated mistakes.
When implemented effectively, this capability shifts supplier engagement from a reactive, manual process to a scalable system. Supplier identification moves from weeks to hours. Onboarding timelines compress significantly. And perhaps most importantly, the organization begins to build a durable knowledge base of supplier performance over time.
The Next Challenge: Execution Without a System of Record
Even when sourcing is streamlined, most organizations encounter a second breakdown once a project begins.
The work of actually managing an external study (e.g., tracking milestones, coordinating with suppliers, managing deliverables) typically happens outside of sourcing or procurement systems. Email threads become the primary communication channel, spreadsheets track timelines, documents are stored across shared drives.
This creates a second layer of fragmentation. Information becomes distributed and communication is difficult to trace. And there is no single, reliable record of what has happened over the course of a project or set of projects.
The consequences are well understood: missed milestones, delayed interventions, incomplete audit trails, and increased coordination overhead across teams.
Capability 2: Project / Study Management
If Marketplace & Supplier Management defines how work enters the system, Project/Study Management defines how it is executed, and whether it remains connected throughout its lifecycle.
A cohesive system provides:
- A unified project workspace
Stakeholders across biopharma R&D, procurement, suppliers, and compliance all operate within the same environment, with access to shared data, communication, and documentation. - End-to-end lifecycle coverage
From RFP and proposal evaluation through execution and final deliverables, every stage of the project is managed within the same system. - Real-time milestone tracking and visibility
Progress is continuously updated, with automated alerts for at-risk timelines, enabling proactive intervention rather than reactive escalation. - Centralized supplier communication
All interactions are captured within the platform, creating a complete, searchable record tied to the project. - Complete auditability
Every action, change, and approval is documented, supporting both internal governance and regulatory compliance.
The impact of this capability is not just improved visibility, it is continuity.
Instead of managing a project across multiple tools, with information stitched together after the fact, organizations operate within a single system of record. Context is preserved. Data flows naturally from sourcing into execution. And every stakeholder works from the same source of truth.
Without a single system of record, external R&D projects fragment across emails, spreadsheets, and shared drives—and the cost is more than inefficiency.Concluding Remarks
For many organizations, external R&D is still managed as a series of transactions, with each supplier engagement, each project, and each payment handled separately.
But as complexity increases, that model becomes increasingly difficult to sustain.
Marketplace & Supplier Management and Project/Study Management represent the first step in moving beyond that model. Together, they transform external R&D from a set of disconnected activities into a continuous, system-driven workflow.
And increasingly, it is a requirement for operating at scale.
Want the full evaluation framework? Download The Enterprise Buyer's Guide to Scientific Operations Software for the detailed requirements, evaluation questions, and vendor scorecard.